|
Today, Andrew decided that today would be a good day for a guided activity. Not everybody needed to participate, but if you wanted to you had the freedom to do so. The reason I chose to participate was that the points in store sounded really appealing, but I was also interested because I thought I could be more engaged in this class and it would help me if I did.
First off, Andrew explained what a chemical reaction was. A chemical reaction is where a change in matter occurs where the atoms in one or more substances combine separate or rearranging to form new things. And these combustion reactions actually power things all over the world, that these things somehow make the energy we need. He explained that molecules are made with the combination of elements/atoms. But, these combine when the amount of both make a full shell of electrons. He showed us a diagram in where there was the nucleus, which was the ball, and energy levels, which were these rings around the nucleus, and then could be a combo of more than two electrons which where dots on the energy levels. He showed us an example, water, and told us why it is written as H2O. I realized this was because oxygen has 6 electrons and needed two more to complete the outer shell, so we needed two more atoms of hydrogen where each of these atoms has 1 electron, so that would complete the shell. As for the experiment itself, we bunched up magnesium strings and put it in vials of citric acid mixed with water. Once we did that, we found that the vial created heat and at the same time, the balloon that we put on top of the vial started filling up. And it is because the magnesium made a chemical reaction with the citric acid and made it create an oxygen, and that heat was the activation energy that released the gas and filled the balloon. Im really glad that i decided. We will be making motors in this class, so I feel learning about chemical reactions and things like this will definitely be useful because chemical reactions also occur during the motor. I felt the way Andrew explained the terms went really well, I found it relatively easy to take notes along the way. I also found it enjoyable how we got to actually take part in the experiment, instead of just having the teachers do the experiment. Overall, while I originally only did the activity to potentially get 50 points, I found it very enjoyable and informative.
0 Comments
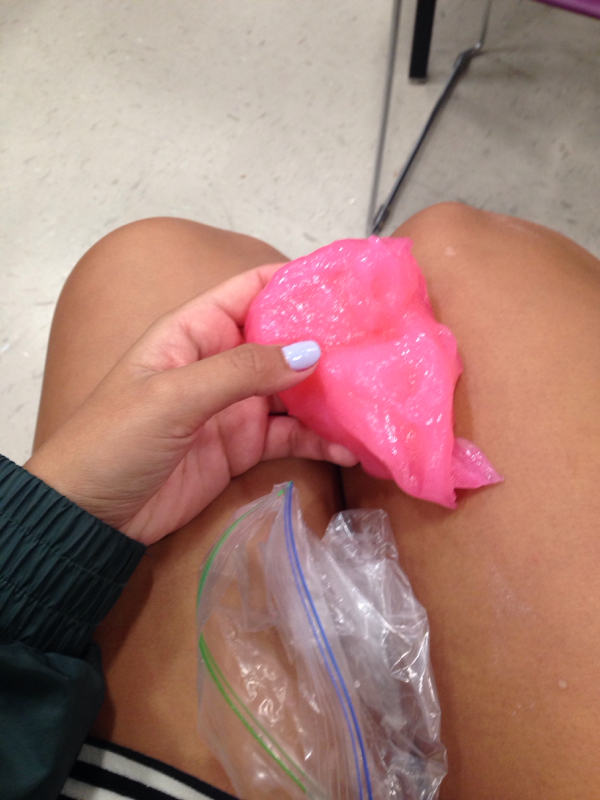
After getting our groups together I found mine. My group members consist of Kobe, Lupe, Nick and I. This week we've had to research and understand what a Core Rocket Motor and an End Burner Rocket Motor is. We also had to research the two different types of fuel like a Potassium Nitrate/Sugar and Black Powder. After discussing and researching, we decided that our best bet would be to go with Potassium/Sugar End Burner Rocket. Today Lupe and I were trying to understand the motors better and we were researching and looking at videos to help us. I came across this video that the pair of us out significantly. Today Noah and I attempted at adding more home made magnetite to our silly putty. Since we made it ourselves we'd have to get some iron and grind it up into a fine powder and then just knead it in. We grabbed a magnet and put it in a bag and then Ina. Shirt and we kept banging it with a hammer so it'd be easier to grind. Then we were grinding it but it was weird. It was a magnet and I was like.. "Noah did you use a magnet last time??..." And he was like "no but it'll b fine I think" I didn't think so but I just let him do him. And then he was like this is fine enough but let me tell you. That thing was just straight lik rocks like it was NOT a powder but on well. He put it in and it looked terrible. And it just started falling a part and it looked nasty. If only we had used iron... But we still had some silly putty left so it wasn't a complete failure! We gained a truckload of knowledge too so!!! Today as I was waiting for Noah to finish his rocket project thing with Alex, I looked over and saw that Isabella was about to do something far out. I asked if I could go out with her and check it out and she let me. it turns out that her and Zion were making Kinetic Sand! When I got outside they had beach sand drying and laid out. We then transferred it into a lil bowl or bucket and measure it. It needed to be like 4 cups I believe maybe 6. The instructions said that we needed a liquid that could water proof clothing to protect them. Looking back I know that we used to actual name but I am not sure what the same for is now... Clothing protector? Anyway, they said that liquid starch would do and I didn't think it would but I was like "eehhhhhh ok I'll let them do them". Now the didn't have liquid starch and Noah had just bought some that day for our silly putty (the right kind for the first time). So I let them use it while I was there we put one cup I think, into the dry sand and they mixed it in together. And they put like 2 cups of water as well. It looked like straight beach sand with water so it didn't work out. This is when I decided to help. So I googled searched what else we could find and I had just hit the jackpot. I found one that needed Dish Soap, Baking Soda and Baking Powder. It didn't say the measurements so we just watched our mixture and the privided photos and videos as a reference. We put the baking soda first, then the powder than some soap. I put everything in the bowl and j was scared to put too much soap so I did it very slowly and let it mix in, in between. The instruction for measurements just said that if you want it "fluffier" you just add more baking power. It took a lot of time to finally get it how we wanted and then we sat and played with it for a while. We then realized that nobody took photos and so then Isabelle tried with her messy hands. After thinking a lot and researching a little I found the science: Kinetic sand is a non-Newtonian fluid (like oobleck) tnat increases viscosity under stress or pressure. With oobleck it's usually liquidy until you punch it. Then it's solid. When you release the stress or pressure it flows like a liquid. Kinetic sand works the same way but it's a lot stiffer and thicker, since after all, it is sand! Today was a very productive and they opposite of a disappointing day! I made kinetic sand and also succeed for the first time in many tried with the silly putty. Noah walked in with a big bottle that clearly said "liquid starch". Of course I was still afraid it wasn't going to work but it did! Noah have me the OK to start a little early and so I did. He was kind of busy with his rocket. So anyway, I got the ingredients and a bowl. So I first put about a handful of elmers all purpose glue into the bowl, and then added like 3 teaspoons of the liquid starch. It started to feel like putty but I could really tell I needed to ads more starch. So I did and mixed it and kneaded it in my hands. I was EXTATIC because Noah and I had tried so many times to make this and we finally got it! It was like legitimate putty! But of course I had to add some pink food coloring because I just live it so darn much! And it was really cool. Noah played with it and showed all his buddies and it was nice. Like celebration and everybody seemed interested. I am super into transparent stuff so I was like hmmmm we have clear glue.... Would this work? It's not all purpose but whatever! What do we have to lose. (The all purpose glue bounced will the others don't, according to our source) I put myself in the corner of the classroom because I knew this was gonna be so awesome And I mixed it all in a measuring cup. It looked really cool but a little weird like something you dont want it to look like. It was thick like putty but cleanish whitish. So I showed my friends and they laughed then I added pink food coloring and it so beautiful!!!!!!!!!! I was in sho k like wow! Then i.e. As like ok I'm gonna use the rest of the glue I don't care. And so I made it. And I put it in the lil baggy. And I mixed the two together and it turned into a slightly lighter pink and I was in love. After that Noah tried adding the magnetite to our first successful mixture that day and ya. So we needed more magnetic and so we thought we would do that Monday considering we didn't have enough to time now. And so we did. The science behind this isn't as clear to me. I think that the glue is just the glue, and the liquid starch keeps it together. And keeps it from sticking to people or things, not at all like our previous attempts.
As a small lab, we looked at sodium polyacrylate. Sodium Polyacrylate is a substance used in diapers, as it can soak up 300 to 800 times its volume of water, or other liquids. This occurs because of the structure of the chemical. All polymers are molecules that are made of small, repeating units that form long chains. In its dry crystal form, sodium polyacrylate is coiled. When water is added, the long chains of sodium polyacrylate stretch out. This happens because each repeating unit in sodium polyacrylate contains an area that can support a negative charge. When water is added to the polymer, these areas form negatively charged ions that repel one another and cause the polymer to stretch out. As the polymer stretches, more water molecules can associate with its areas of negative charge.
We started with a measured amount of sodium polyacrylate, taken from diapers, in a glass jar. Then Braden stirred in water gradually, causing the chemical to swell. By the end, the jar was almost completely filled! Braden said it was possible to cause the jar to overflow, but it would be a mess to clean up. The gel that it created was almost like a warm snow but squishy and dry. |



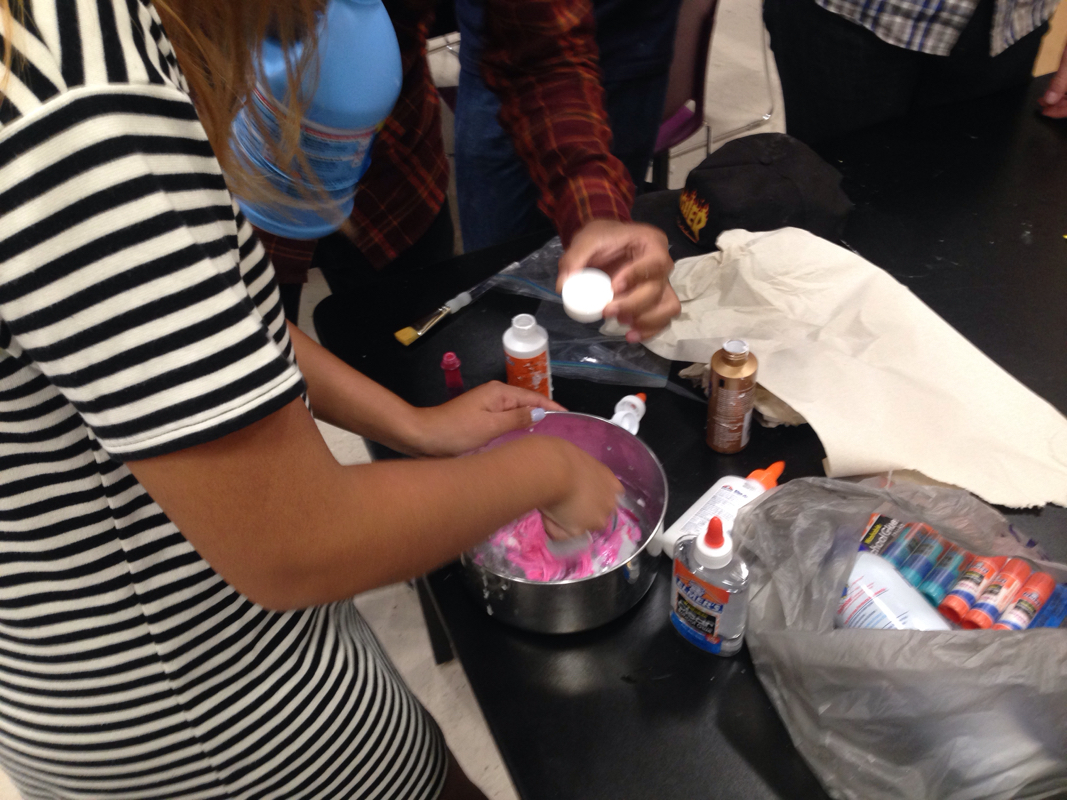




 RSS Feed
RSS Feed
